|
3/17/2024 0 Comments Electron region geometry chart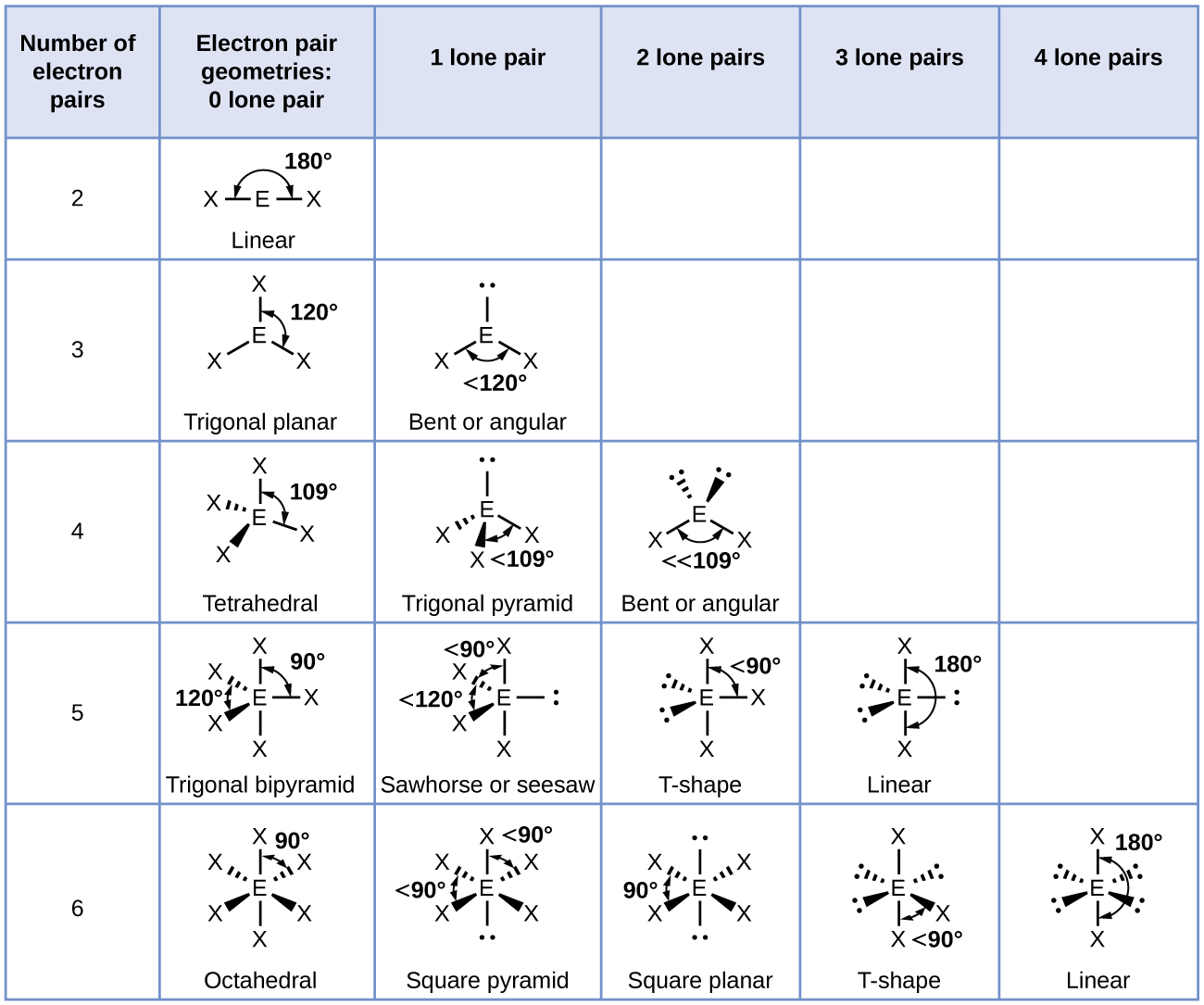
As we discussed the formula to calculate the formal charge earlier, it can be done as follows īonding electrons are the ones that are involved in bond formation. The following image shows the lewis structure which has all the bonds and lone pairs visible along with the satisfied octet rule.įor completing the lewis diagram, the last step is to calculate the formal charge. We know that two bonding electrons together form a bond which is represented by a line. The following shows how the valence electrons are placed around the respective atoms.Ĥ) Satisfy the octet rule of each atom and sketch the final lewis structureĪs there are a total of 14 valence shell electrons in methanol, therefore, there must be 7 electron pairs in the molecule. The rough sketch looks something like the following image.ģ) Placing the valence electrons around individual atoms As we can observe, methanol has three C-H bonds, one C-O bond, and one O-H bond. Here, we will be moving further by taking carbon as the central atom. However, the lewis structure of methanol can be drawn easily by considering either of the elements as the central atom. Therefore in methanol, both C and O are appropriate central atoms. The central atom must not be strongly electronegative and must not be too small in size. (1*4) + (1*6) + (1*4) = 14 valence shell electronsįor drawing a rough sketch, it is required to select the central atom of the molecule. Therefore the total valence electrons are: 
Now, there is 1 carbon atom, 1 oxygen atom, and 4 hydrogen atoms in one CH3OH molecule. Take a look at the following table for the same. of valence electrons in a molecule is the sum of valence shell electrons of each element in the molecule. Steps to Draw the Lewis Structure of Methanol (CH3OH)ġ) Calculation of total valence electrons in methanol 
It is calculated by the following formula įormal charge of an element = (Valence electrons) – ((bonding electrons)/2) – (Non bonding electrons) The sum of these formal charges has to be equivalent to the net charge on the molecule. This is because noble gases are considered to be stable in nature.Ī formal charge is a theoretical charge present on elements of a molecule. The octet rule states for every element to obtain a noble gas configuration i.e to have a total of 2 electrons (in the case of H and He) or a total of 8 electrons in their valence shells. Here, the valence electrons are represented by small dots and since a single bond consists of two bonding electrons, the two dots between two atoms are represented by a line instead, which represents a bond between them.Ī lewis structure gives us information about the existence of a bond between atoms, although it is not enough to tell us about the type of the bond.įor making the lewis structure of methanol, it is required to satisfy the octet rule first and then calculate the formal charge of every element in the molecule. The Lewis Structure of a molecule gives the simplest representation of valence shell electrons around itself.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
